
A vault particle. Image adapted from Tanaka et al. on Wikimedia Commons under CC BY SA-3.0.
In 1986, researcher Nancy Kedersha and her supervisor Leonard Rome sat in the basement of the UCLA Biological Chemistry Department, next to an intimidating electron microscope that towered over the rest of the room.
After several days of meticulously preparing their model of choice — rat liver cells — they transferred their samples to a thin carbon film, stained them, and stared down the electron microscope. What they saw were gigantic (by cellular standards, at least) protein structures formed of multiple arches that tapered at both ends. They named them vaults, after the arched structures in cathedral ceilings.
They noted that:
[t]he function of vaults is at present unknown
Kedersha and Rome, Journal of Cell Biology, 1986
and since 1986, this has remained the dominant view. They’re the largest particles human cells produce, and there are thousands of them in each cell, but we still don’t have a clue what they’re for. They’re found in other mammalian cells too, so are clearly vital.
Papers have found that vaults can store RNA, particularly messenger RNA (mRNA), which is the cell’s instruction manual for printing proteins. But the reason why the cells need to do this remained uncrackable, and both momentum and funding slowly dried up.
Now, 40 years on from Kedersha and Rome’s paper, a small team led by Yu-Kai Chao at the Broad Institute in the US created artificial vaults, and found that they act as tiny time capsules to record and store the transcriptional output of our cells, providing some insight into this decades-old question.

Creating the vaults
To mimic naturally occurring vaults, the authors engineered protein particles which could self-assemble. These proteins were called TimeVaults, which have a similar structure to the real thing. They could capture mRNAs from the cytoplasm and store them, a little like catching fish in a pond. TimeVaults caught mRNAs around 20 times more efficiently than other similar mRNA storage systems (like those in bacteria).

Importantly, after cell division the TimeVaults were still present at a similar distribution in the cells, suggesting these engineered structures are stable throughout generations.
Don’t shoot the messenger
But was the messenger RNA inside them stable?
After lysing cells (removing their membranes, and just keeping the material inside) containing TimeVaults and then treating them with RNase, an enzyme which degrades RNA, they found that mRNA stored within TimeVaults was stable for at least 8 days. In contrast, lysates from cells without TimeVaults saw a much earlier drop-off of mRNA. TimeVaults therefore acted as a molecular time capsule, shielding mRNA from degradation.
Introducing such large proteins into cells is no mean feat, and so the authors wanted to ensure they didn’t impact cellular function. They performed RNA-seq to investigate whether TimeVaults warp the global gene expression of the cell, and found no statistically significant impact. This is important for those who may want to use TimeVault constructs in future for recording transcripts (although the impact may vary depending on cell type).
Putting TimeVaults to the test
The authors applied two types of stress (heat shock and hypoxia) to their cells to see whether TimeVaults kept any record of stress-related transcriptional changes.
One of these stresses was hypoxia, or insufficient oxygen. Intriguingly, more mRNAs associated with hypoxia response were locked away in the TimeVaults compared to before the stress, suggesting these vaults can indeed store transcriptional “memories” of stress.
Clinical applications
The authors also tested whether TimeVaults captured different mRNAs when treating cancer cells with a growth blocker drug. For this, they tested two classes of PC9 lung cancer cells, one which would persist after drug treatment and one which was more sensitive.
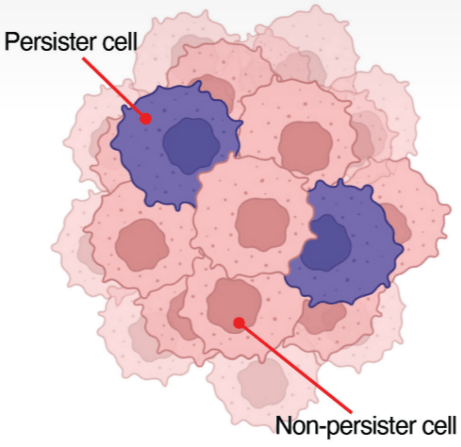
They found that in persister cells, TimeVaults recorded an upregulation of genes involved in metabolism (particularly oxidative phosphorylation, our body’s main programme of generating energy), which is consistent with previous reports of drug-resistant cancer cells relying on this programme more for survival.
“Yeah, and?”
OK, but why is knowing about vaults useful?
Firstly, it allows us to get one step closer to real-time recording of transcription in our cells, which is arguably one of the biggest problems in modern day molecular biology. Current experimental approaches such as RNA fluorescent labelling or RNA sequencing both only capture snapshots of a cell’s RNA profile, but can’t track anything live as it happens.1
Secondly, it may provide clues as to why cancer cells may be resistant to certain drugs. TimeVaults’ ability to create a time capsule of past mRNA transcripts may help us identify which transcripts need to be targeted in future therapies.
And finally, it brings us closer to finding out the real function of the mysterious structures Kedersha and Rome saw down the microscope 40 years ago.

Notes
- There is a computational technique to measure RNA expression over time, which is called RNA velocity. RNA velocity uses statistical models to predict which RNAs a group of cells will “print” out over time. However, these models can make assumptions, meaning RNA velocity predictions don’t always match up with experimental results. For more details, see this paper. ↩︎
Discussion point
Besides their clinical implications and ability to record stress within cells, what other applications could this method of storing mRNA have?


Leave a comment